Zusätzliche Informationen
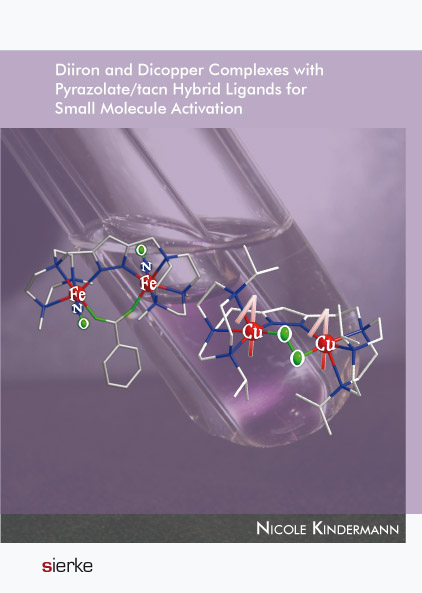
| Das Buch | The presence of metal-containing enzymes is ubiquitous in nature. Besides structural stability that metals might exert on the protein backbone, their Lewis acidity and/or the redox activity provide an excellent basis to perform catalytic transformations of all kinds. In order to unveil the underlying mechanisms, low molecular weight analogues have been developed by bioinorganic chemists and studied by various techniques. The focus of this dissertation is the development of novel dinuclear iron and copper complexes as bioinspired models for non-heme diiron and type-3 copper proteins. These proteins contain dinuclear centers in the respective active site, and are known to activate small molecules such as dioxygen or nitric oxide. Using ligand scaffolds based on pyrazole and triazacyclononane (tacn), the isolation of diiron and dicopper complexes is achieved, mimicking biologically relevant metallosites in biomolecules. A detailed analysis of the compounds by X-ray diffraction and by various spectroscopic techniques, e.g. UVvis, FTIR, (resonance) Raman and Mössbauer spectroscopy, is given. In addition, their reactivity towards small molecules, the characterization of adducts and their further transformation, i.e. by redox or acid-base chemistry, are described. |
|---|